Introduction
Myeloproliferative neoplasms are considered mutually exclusive, and reports of co-occurring mutations are rare. Over the past 10 years, approximately two dozen case reports have been published of co-occurring BCR-ABL positive and CALR positive patients. Here we present the case of a patient who was reported to have myelofibrosis (MF)/myeloproliferative neoplasm (MPN), later developed chronic myeloid leukemia (CML), and ultimately progressed to acute myeloid leukemia (AML). This is unique in the literature reviewed, and may be the first reported case of this overlap syndrome being treated with asciminib.
Case presentation
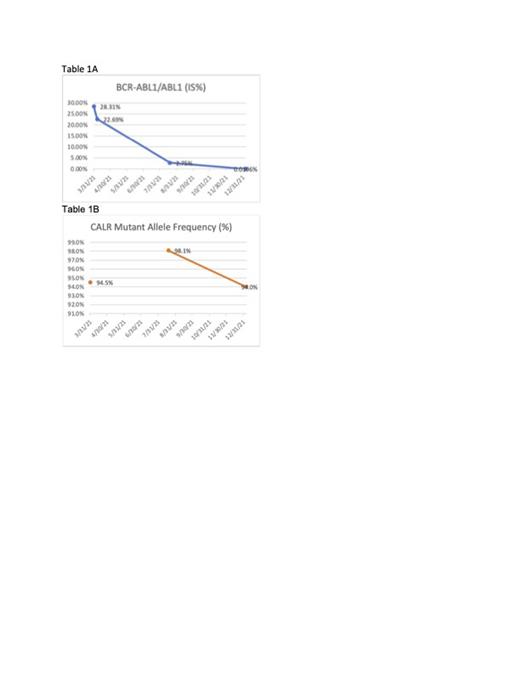
A 40-year old male was diagnosed in 2018 with MF/MPN based upon a hypercellular bone marrow biopsy with diffuse reticulin fibrosis. He was negative for JAK2, but was not tested for CALR or MPL mutations. Treatment was ruxolitinib and hydroxyurea. Two years later, the patient was diagnosed with CML after being admitted for recurrent fevers, headache, and shortness of breath. At that time, a type 2 CALR mutation (CALR K385Nfs*47) was noted. He started dasatinib and decitabine, which resulted in a drop from 28.31% BCR-ABL to 0.0196% (Table 1A). However, hyperuricemia and renal failure led to a change in regimen and the patient was started on asciminib and ropeginterferon alfa-2b-njft in January 2022. By December, the patient's BCR-ABL PCR dropped below quantification, but he received a diagnosis of AML based on a bone marrow biopsy which showed 43% blasts. He received cladribine and cytarabine, but was found to have leukemic infiltration of the spinal cord five months later. The patient was referred to hospice and passed away in June 2023.
Discussion
To our knowledge, this is the first MPN-CML patient reported to have been treated with asciminib. This case illustrates its effectiveness in treating CML, but highlights the need for improved treatments for CALR+ MPN. Moreover, this case illustrates the importance of testing for CALR, MPL, and BCR-ABL in addition to JAK2 immediately when a patient presents with an MPN.
It is unknown whether this patient's CALR mutation preceded the BCR-ABL translocation, and there is debate in the literature with respect to mutation chronology. The patient's BCR-ABL burden dropped from 28.31% to 0.0196% while the CALR burden remained around 94% over the same period (Table 1B). Thus, it is unlikely that the BCR-ABL mutation preceded the CALR mutation within the same clone.
The patient's >90% CALR allele burden is higher than reported in the other reported cases, where the burden ranged from 10% to 68%. Furthermore, none of the other cases were reported to progress to AML, though follow up data is lacking. The heavy allelic burden in our patient may have been a contributing factor to the development of AML, as has been reported elsewhere.
The patient's overall mutational profile is of interest. Type 2 CALR mutations have been reported to have less favorable prognoses than type 1 mutations. In addition to the CALR and BCR-ABL mutations, the patient had ASXL1 A861Hfs*6, EZH2 D730*, and NRAS G12V. All three mutations are associated with unfavorable outcomes in MDS, AML, and/or CML. Genomic instability has been discussed in relation to both CML and JAK2-positive MPN. Our patient's heavy mutational burden suggests CALR-positive neoplasms may also present against a background of inherent genomic instability.
Conclusion
We reported a case of MPN, followed by CML, and then AML. The patient was positive for CALR and BCR-ABL. He achieved a complete response in CML after targeted treatment, with asciminib consolidating the gains the patient made on earlier therapies. However, he always maintained a high CALR burden. This patient's mutational profile and disease course argue for the importance of early testing for CALR and BCR-ABL in addition to JAK2 and MPL in the setting of an MPN and for further research into the underlying causes of these overlap conditions.
Disclosures
Seiter:Rigel: Membership on an entity's Board of Directors or advisory committees; Abbvie: Membership on an entity's Board of Directors or advisory committees; BMS: Membership on an entity's Board of Directors or advisory committees; CTI: Speakers Bureau; Incyte: Speakers Bureau; Alexion: Honoraria; Servier: Speakers Bureau; Novartis: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Blueprint: Speakers Bureau. Liu:Beigene: Speakers Bureau; Rigel: Speakers Bureau. Steinberg:MorphoSys: Membership on an entity's Board of Directors or advisory committees; Jazz Pharmaceuticals: Speakers Bureau.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal